Pregnancy-Safe Blood Pressure Medication Checker
Medication Safety Check
Select the medication you are currently taking or considering. This tool provides general information based on current medical guidelines regarding pregnancy safety.
Recommendations:
If you are taking medication for high blood pressure and find out you are pregnant, the clock starts ticking. It is not just about managing your health; it is about protecting the developing kidneys of your baby. ACE inhibitors and ARBs are two common classes of antihypertensive drugs that block the renin-angiotensin system to lower blood pressure. While they are standard care for millions of people worldwide, they carry a strict warning label for pregnancy: do not use them.
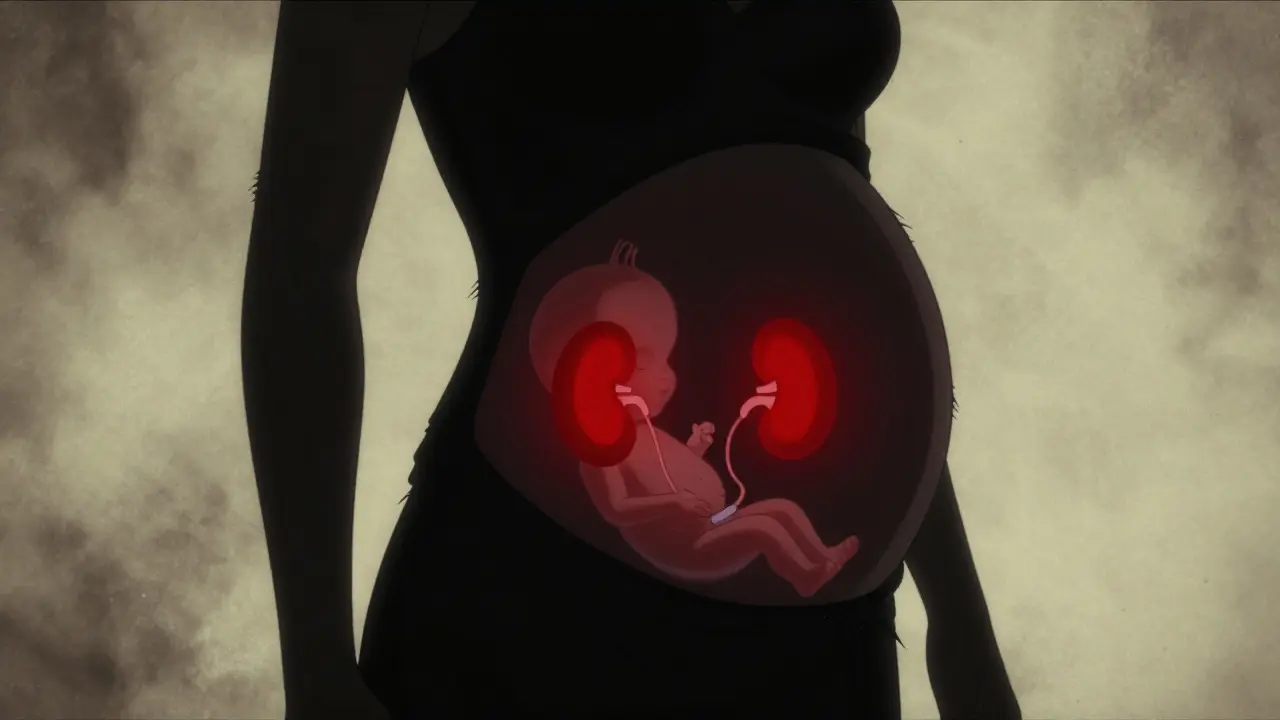
The reason is biological, not theoretical. Your baby’s kidneys rely on the very system these drugs block to produce urine and amniotic fluid. When you take an ACE inhibitor or an ARB during pregnancy, you can inadvertently shut down this process, leading to severe complications like kidney failure in the fetus, dangerously low amniotic fluid (oligohydramnios), and even skull defects. This article breaks down exactly why these drugs are dangerous, what happens if you have already taken them, and which safer alternatives your doctor will likely switch you to.
Why ACE Inhibitors and ARBs Are Dangerous for Fetuses
To understand the risk, we need to look at how these medications work. Both ACE inhibitors (like lisinopril or enalapril) and ARBs (like losartan or candesartan) target the Renin-Angiotensin-Aldosterone System (RAAS). In adults, RAAS helps regulate blood pressure by controlling fluid balance. In a developing fetus, however, RAAS is critical for kidney development and function.
When a pregnant person takes these medications, the drug crosses the placenta. The fetus’s kidneys cannot produce urine effectively because the angiotensin II pathway is blocked. Without urine production, amniotic fluid levels drop drastically. This condition is called oligohydramnios. Amniotic fluid protects the baby and allows room for movement; without it, the lungs may not develop properly, and the umbilical cord can become compressed, cutting off oxygen.
The consequences can be severe and sometimes fatal. According to data from Medsafe (New Zealand’s medicines regulator) and studies published in journals like *Obstetrics & Gynecology International*, exposure to these drugs is linked to:
- Fetal renal damage: Permanent scarring or failure of the kidneys.
- Oligohydramnios: Critically low amniotic fluid.
- Skeletal defects: Including skull malformations due to compression in the womb.
- Hypotension: Dangerously low blood pressure in the newborn.
- Hyperkalemia: High potassium levels in the blood, which can affect heart rhythm.
- Fetal death: In severe cases, the combination of organ failure and lack of fluid leads to miscarriage or stillbirth.
Research by Moretti et al. (2011) highlighted that while major structural birth defects might not always occur, babies exposed to these drugs had significantly lower birth weights (about 350g less on average) and were born earlier (1.8 weeks early). More alarmingly, the rate of miscarriage was more than double in the exposed group compared to controls (25.4% vs 12.3%).
Is First-Trimester Exposure Safe? The Controversy
You might have heard conflicting advice. Some older studies suggested that first-trimester exposure was relatively safe regarding major physical deformities. However, medical consensus has shifted dramatically. A 2020 meta-analysis by Buawangpong confirmed that first-trimester exposure is associated with increased risks of adverse pregnancy outcomes. The American Heart Association (AHA) notes that while the risk of major malformations is debated, the risk is "not negligible," and the danger increases substantially as the pregnancy progresses into the second and third trimesters.
There is also evidence that ARBs may pose a higher risk than ACE inhibitors. The AHA’s 2012 review specifically stated that neonatal outcomes are poorer following prenatal exposure to ARBs. Because the risks are so significant and potentially irreversible, major health organizations treat all trimesters as high-risk periods. There is no "safe" window for using these drugs during pregnancy.

Immediate Steps If You Are Pregnant or Planning To Be
If you are currently taking an ACE inhibitor or an ARB and discover you are pregnant, do not panic, but act quickly. The guidelines from the American College of Obstetricians and Gynecologists (ACOG) and Te Whatu Ora - Health New Zealand are clear: discontinue the medication immediately upon confirmation of pregnancy.
Here is what typically happens next:
- Contact your healthcare provider: Inform them of your pregnancy status right away. Do not stop your medication without professional guidance if your blood pressure is critically high, but expect a rapid transition plan.
- Switch to a safer alternative: Your doctor will likely prescribe a different class of medication that does not interfere with fetal kidney development.
- Monitor closely: You will likely undergo detailed ultrasounds to check amniotic fluid levels and fetal growth, especially if exposure occurred in the second or third trimester.
If you are planning to become pregnant, the advice is even stricter. Medsafe’s 2024 advisory mandates that providers exclude pregnancy before starting treatment and counsel patients on the risks. If you are on these meds, talk to your doctor about switching to a pregnancy-safe option before conception. This proactive step eliminates the risk entirely.
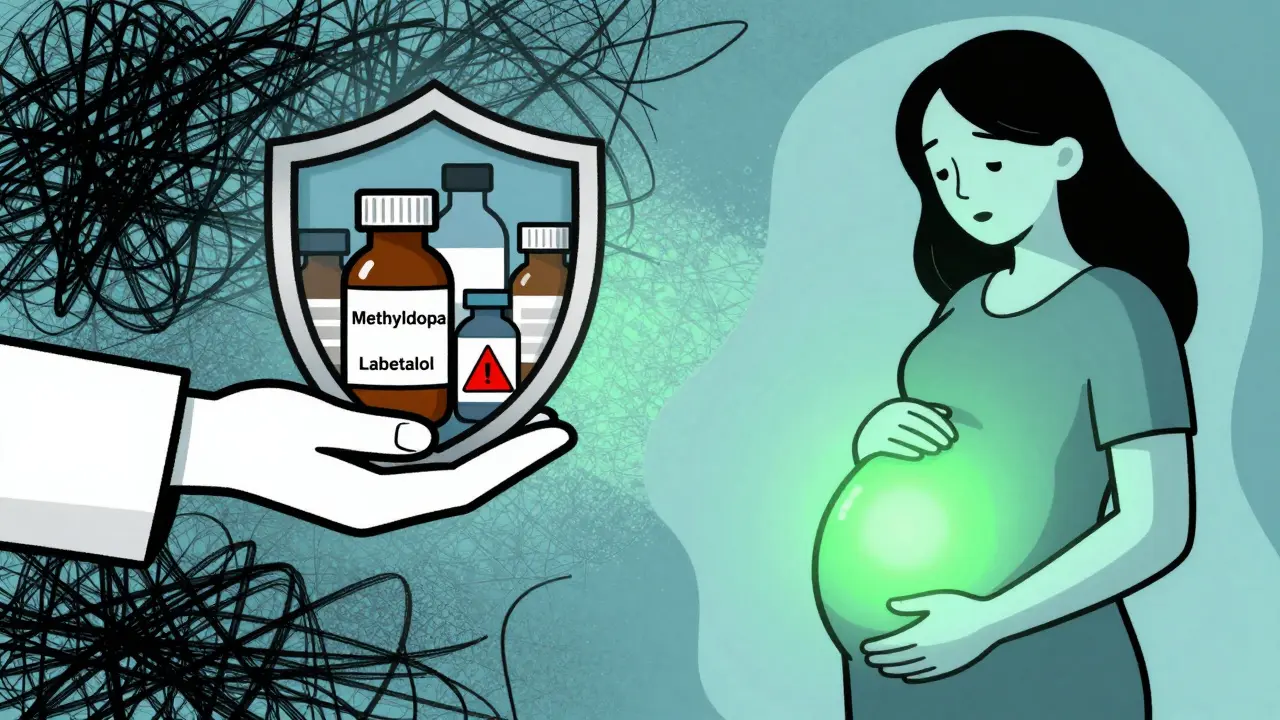
Safe Alternatives for Managing Hypertension in Pregnancy
High blood pressure during pregnancy is serious and must be treated. Untreated hypertension can lead to pre-eclampsia, stroke, or placental abruption. Fortunately, there are effective medications that are considered safe for both mother and baby. The choice depends on your medical history, but three main options dominate clinical practice.
| Medication | Class | Role in Pregnancy | Key Considerations |
|---|---|---|---|
| Methyldopa | Centrally acting alpha-2 agonist | First-line therapy | Longest safety record (since 1970s). Minimal side effects for the fetus. Can cause drowsiness or dry mouth in the mother. |
| Labetalol | Beta-blocker | First-line / Alternative | Dual action (alpha and beta blockade). Very effective. Preferred by many clinicians due to better maternal tolerance than methyldopa. |
| Nifedipine | Calcium channel blocker | Second-line therapy | Used if other options fail. Caution needed in women with certain heart conditions. Effective for acute spikes in BP. |
Methyldopa has been the gold standard for decades. Studies dating back to the 1970s show no increase in birth defects or long-term developmental issues in children exposed to it in utero. It works by calming the signals from the brain that raise blood pressure. Starting doses are usually low (e.g., 250mg twice daily) and titrated up to a maximum of 3g per day.
Labetalol is increasingly favored as a first-line treatment. It blocks both alpha and beta receptors, providing strong blood pressure control with fewer side effects for the mother compared to methyldopa. Typical starting doses are around 100mg twice daily, adjustable up to 2,400mg/day depending on response. It is particularly useful for preventing pre-eclampsia-related complications.
Nifedipine is often used as a second-line agent or for managing acute hypertensive crises. It is a calcium channel blocker that relaxes blood vessels. While generally safe, it should be used with caution in patients with existing cardiac disease due to potential negative effects on heart muscle contraction.
Regulatory Warnings and Clinical Guidelines
The medical community is united on this issue. The U.S. Food and Drug Administration (FDA) previously categorized these drugs as Pregnancy Category D (positive evidence of human fetal risk). Current labeling includes a boxed warning-the strongest type-alerting prescribers to the risk of fetal toxicity. Similarly, the European Medicines Agency (EMA) and the World Health Organization (WHO) exclude ACE inhibitors and ARBs from their lists of essential medicines for pregnancy.
In New Zealand, Te Whatu Ora - Health New Zealand’s 2024 clinical guideline explicitly states that ACE inhibitors and ARBs should be discontinued prior to conception or as soon as pregnancy is confirmed. They recommend methyldopa as the primary choice, with labetalol as a close alternative. Blood pressure targets for pregnant women without end-organ damage are typically set below 140/90 mmHg.
Despite these clear warnings, errors happen. FDA data from 2021 showed that 1.2% of pregnancies in women with chronic hypertension still involved exposure to ACE inhibitors or ARBs. This highlights the importance of preconception counseling. If you are of childbearing age and prescribed these medications, ensure your doctor discusses contraception and the need to switch drugs if you plan to conceive.
What If I Accidentally Took These Drugs?
If you took an ACE inhibitor or ARB for a short time before realizing you were pregnant, the risk depends on the timing and duration. Early exposure (first few weeks) may result in an "all-or-nothing" outcome, meaning either the pregnancy is lost naturally or the embryo continues without major impact. However, exposure beyond the first trimester carries a much higher risk of renal damage and oligohydramnios.
Do not self-diagnose or guess. If you suspect exposure, your obstetrician will likely order detailed anatomy scans and Doppler ultrasound studies to monitor fetal kidney function and amniotic fluid volume. Early detection of problems allows for closer monitoring and, in some cases, interventions to improve outcomes.
Can I continue taking Lisinopril if I am trying to get pregnant?
No. You should not take Lisinopril or any ACE inhibitor while trying to conceive. These drugs are contraindicated in all stages of pregnancy. Talk to your doctor about switching to a safer alternative like Labetalol or Methyldopa before you start trying.
Are there any natural ways to lower blood pressure during pregnancy?
While lifestyle changes like reducing sodium intake, staying hydrated, and gentle exercise can help support blood pressure management, they are rarely enough on their own for chronic hypertension. Never replace prescribed medication with natural remedies without consulting your doctor, as uncontrolled high blood pressure poses a greater risk to you and your baby than the medication itself.
What is the difference between ACE inhibitors and ARBs?
Both classes lower blood pressure by targeting the Renin-Angiotensin-Aldosterone System (RAAS). ACE inhibitors block the enzyme that creates angiotensin II, while ARBs block the receptor that angiotensin II binds to. Despite the different mechanism, both are strictly avoided in pregnancy due to similar risks of fetal kidney damage and low amniotic fluid.
Is Labetalol safe for breastfeeding?
Yes, Labetalol is generally considered compatible with breastfeeding. Only small amounts of the drug pass into breast milk, and it is unlikely to affect the infant. Always discuss any concerns with your pediatrician or lactation consultant.
How quickly do ACE inhibitors leave the body after stopping?
Most ACE inhibitors have a relatively short half-life. For example, Lisinopril is typically cleared from the body within 24 hours. However, the physiological effects on blood pressure regulation may take longer to stabilize. Switching to a new medication ensures continuous protection for your baby.
What are the signs of oligohydramnios?
Oligohydramnios often has no obvious symptoms for the mother. It is primarily detected through ultrasound measurements of the amniotic fluid index (AFI). In severe cases, you might notice a decrease in fetal movement or a smaller-than-expected fundal height (the size of your belly).